Neurogenesis is the process by which new neurons are formed in the brain through pre-natal development and as adults.
This phenomenon primarily occurs in the hippocampus playing a crucial role in learning, memory, and cognitive flexibility.
Factors like exercise, enriched environments, and certain drugs can promote neurogenesis, while stress and aging may inhibit it.

Key Takeaways
- Neurogenesis is the formation of new neurons, mostly in the hippocampus.
- It supports memory, learning, mood, and brain repair.
- Although it declines with age, lifestyle factors can boost it.
- Ongoing research aims to harness neurogenesis to treat neurological and psychiatric conditions.
Early Neurogenesis
Early neurogenesis begins during embryonic development when the neural plate – a flat sheet of specialized cells that will become the nervous system – separates from the ectoderm, which is the outermost of the three primary tissue layers in the early embryo and gives rise to skin and nervous tissue.
This separation occurs through a folding process that forms the neural groove.
The groove then fuses to create two key structures: the neural tube, which develops into the central nervous system, and the neural crest, a temporary group of cells.
Neural crest stem cells emerge from this structure and differentiate into various cell types that contribute to tissue and organ development.
During this early stage, neural stem cells differentiate into specialized cell types at specific times and locations within the developing brain, establishing the foundation for the diverse array of neurons that will populate the mature brain.
Where Does Neurogenesis Happen?
In adults, neurogenesis primarily occurs in two brain regions:
- The subgranular zone (SGZ) of the hippocampus: Produces around 700 new neurons per day, helping support memory and learning.
- The subventricular zone (SVZ) near the brain’s lateral ventricles: Sends new neurons to the olfactory bulb, which processes smells.
Emerging research suggests neurogenesis may also occur in the amygdala, a region linked to emotional memory, though this finding is still being explored.
Why is Neurogenesis Important?
New neurons play a critical role in brain plasticity—the brain’s ability to adapt and reorganize.
In the hippocampus, they help form new memories and enhance cognitive flexibility. In the amygdala, they may support emotional processing.
Neurogenesis also offers hope for brain repair. Animal studies show that after brain injury, neurogenesis increases, potentially aiding recovery.
Scientists are investigating how this could be harnessed to treat conditions like Alzheimer’s disease, depression, and traumatic brain injury.
How Does Neurogenesis Occur?
The Process of Neural Development
Neurogenesis begins when neurogenic signals trigger the activation of neural stem cells.
These signals can arise from various sources, including stimulated brain activity and environmental factors.
Once activated, neural stem cells either divide to maintain the stem cell population or differentiate into intermediate neural progenitor cells that are primed for maturation.
These progenitor cells then develop into either neurons or supporting glial cells, depending on the specific signals they receive.
Stages and Integration
As new neurons develop, they grow axons and dendrites—the branches that allow them to connect with other neurons.
These newly formed neurons must then survive and successfully integrate into existing brain circuits. This integration phase is crucial; if the new neurons fail to establish proper connections, they may undergo programmed cell death.
Successful integration allows these neurons to contribute to brain plasticity and adaptation.

Hippocampal Neurogenesis and Memory
In the hippocampus, astrocytes (a type of supporting brain cell) produce specific proteins that trigger neurogenesis.
Research has revealed that approximately 700 new neurons are born daily in the hippocampus, with about one-third of hippocampal neurons being replaced over a lifetime.
This continuous renewal process plays a vital role in forming new memories, but it can also affect existing memories in interesting ways.
Memory Storage and Transfer
When new neurons form in the hippocampus, they can temporarily disrupt memories already stored there.
This happens because memories initially form in the hippocampus but gradually transfer to other brain regions for long-term storage—a process that can take several years.
During this transfer period, the formation of new neurons may weaken existing memories that haven’t yet fully transferred, which might explain why we struggle to retain all memories from our early years.
How Our Understanding Has Evolved
For much of the 20th century, scientists believed neurogenesis ended after childhood.
Early findings by Altman and Das (1965) and later by Paton and Nottebohm (1984) hinted at adult neurogenesis in animals, but these were largely dismissed.
It wasn’t until the 1990s that adult neurogenesis gained widespread acceptance.
Researchers like Richards, Kilpatrick, and Bartlett (1992) discovered neural stem cells in adult mouse brains, proving that even mature brains could generate new neurons.
The Future of Neurogenesis Research
Understanding neurogenesis could lead to new treatments for cognitive decline and mental illness. Scientists are exploring:
- Activating dormant stem cells
- Transplanting neural cells into damaged areas
- Developing drugs that promote neurogenesis safely in humans
While most studies are still in early stages, the potential is promising.
Does Neurogenesis Decline With Age?
Yes, but it doesn’t stop completely. Neurogenesis declines gradually throughout life. In fact, about one-third of hippocampal neurons are replaced over a lifetime. Several factors can slow neurogenesis:
- Aging
- Chronic stress
- Poor sleep
- High blood sugar
- Sedentary lifestyle
Mental health conditions like depression and anxiety can also reduce neurogenesis.
Can You Boost Neurogenesis?
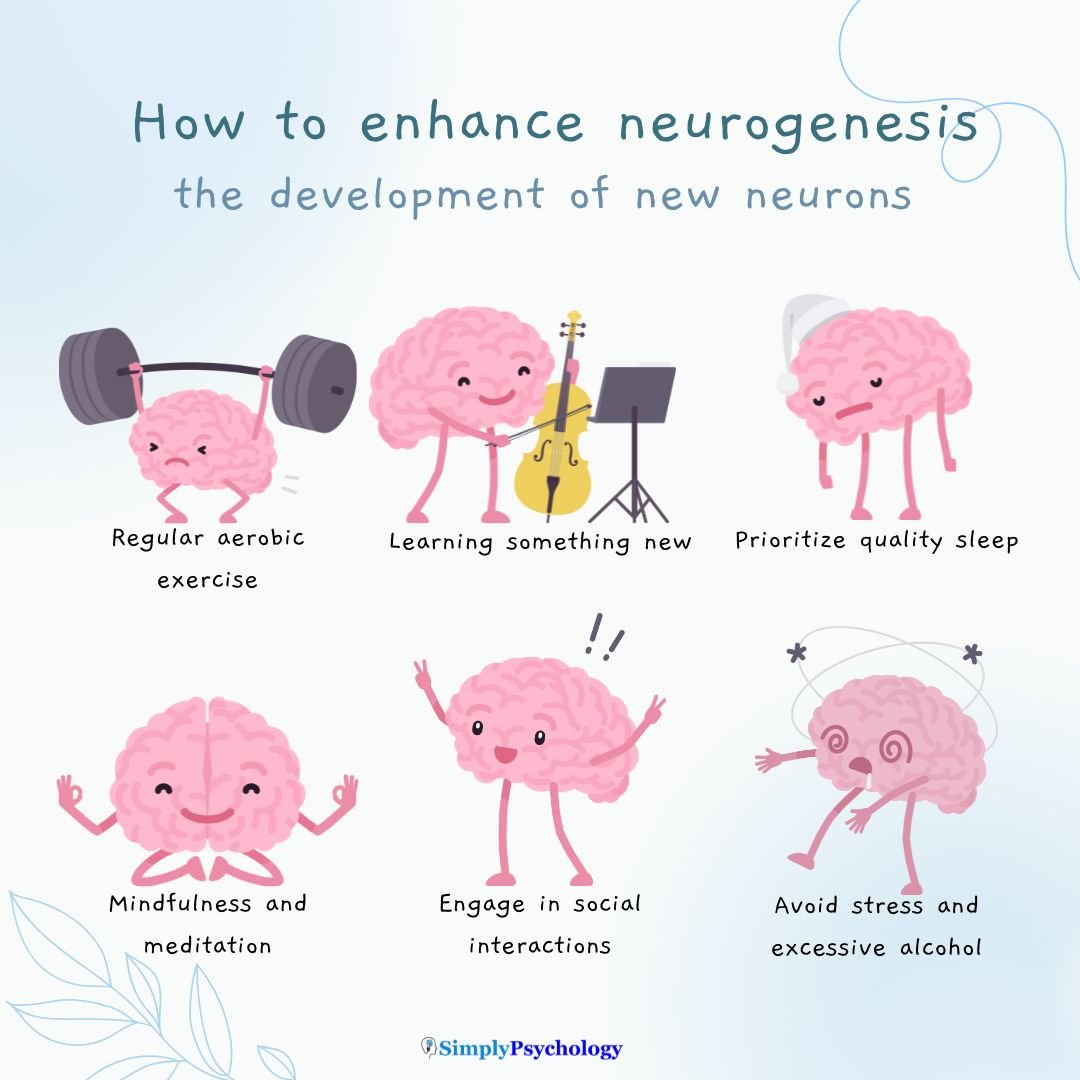
While we can’t reverse aging, certain habits can support neurogenesis at any age:
1. Physical Activity
- Aerobic exercise (like walking or cycling) boosts new neuron growth in the hippocampus.
2. Mental Stimulation
- Learning new skills, solving puzzles, or studying challenging material stimulates brain plasticity.
3. Nutrition
- Diets rich in flavonoids (e.g., blueberries, dark chocolate, green tea) support brain cell growth.
4. Sleep
- Quality sleep is essential for memory consolidation and neural repair.
5. Stress Reduction
Mindfulness, yoga, and relaxation techniques help lower cortisol, which otherwise inhibits neurogenesis.
References
Godos, J., Castellano, S., Galvano, F., & Grosso, G. (2019). Linking omega-3 fatty acids and depression. In Omega fatty acids in brain and neurological health (pp. 199-212). Academic Press.
Götz, M., & Huttner, W. B. (2005). The cell biology of neurogenesis. Nature reviews Molecular cell biology, 6 (10), 777-788.
Kempermann, G., & Gage, F. H. (2000, October). Neurogenesis in the adult hippocampus. In Neural Transplantation in Neurodegenerative Disease: Current Status and New Directions: Novartis Foundation Symposium 231 (Vol. 231, pp. 220-241). Chichester, UK: John Wiley & Sons, Ltd.
Ming, G. L., & Song, H. (2011). Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron, 70 (4), 687-702.
Mira, H., & Morante, J. (2020). Neurogenesis From Embryo to Adult–Lessons From Flies and Mice. Frontiers in Cell and Developmental Biology, 8 .
Queensland Brain Institute (n.d.). What is neurogenesis? Retrieved June 233, 2021, from https://qbi.uq.edu.au/brain-basics/brain-physiology/what-neurogenesis
Queensland Brain Institute. (2017, August 15). Emotion processing region produces new adult brain cells . https://qbi.uq.edu.au/article/2017/08/emotion-processing-region-produces-new-adult-brain-cells
Richards, L. J., Kilpatrick, T. J., & Bartlett, P. F. (1992). De novo generation of neuronal cells from the adult mouse brain. Proceedings of the National Academy of Sciences, 89 (18), 8591-8595.
Rugnetta, M. (2008, July 9). Neural stem cell. Encyclopedia Britannica. https://www.britannica.com/science/neural-stem-cell
Spalding, K. L., Bergmann, O., Alkass, K., Bernard, S., Salehpour, M., Huttner, H. B., Boström, E., Westerlund, I., Vial, C., Buchholz. B. A., Possnert, G., Mash, D. C., Druid, H. & Frisén, J. (2013). Dynamics of hippocampal neurogenesis in adult humans. Cell, 153 (6), 1219-1227.


